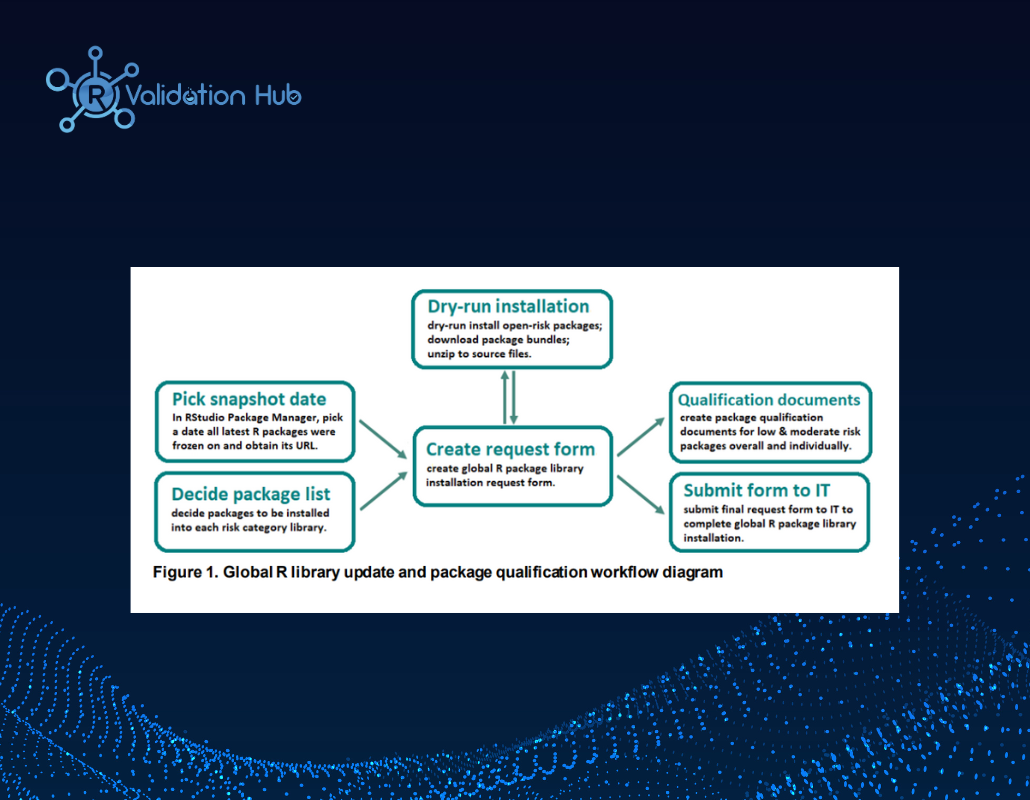
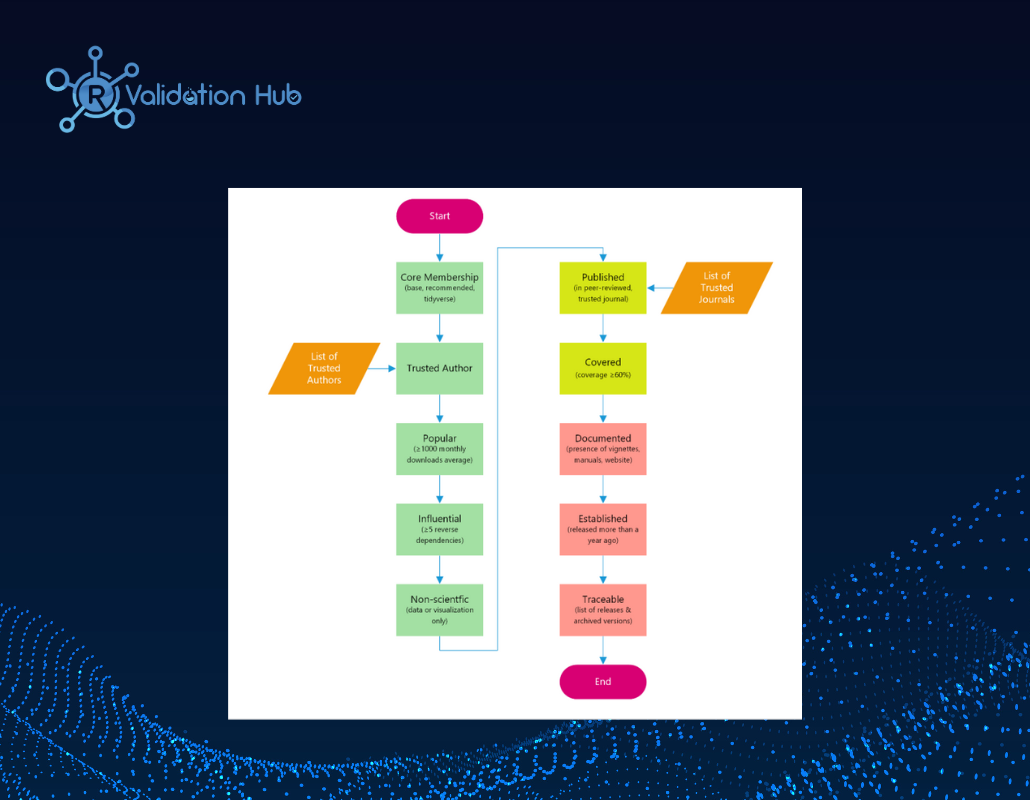
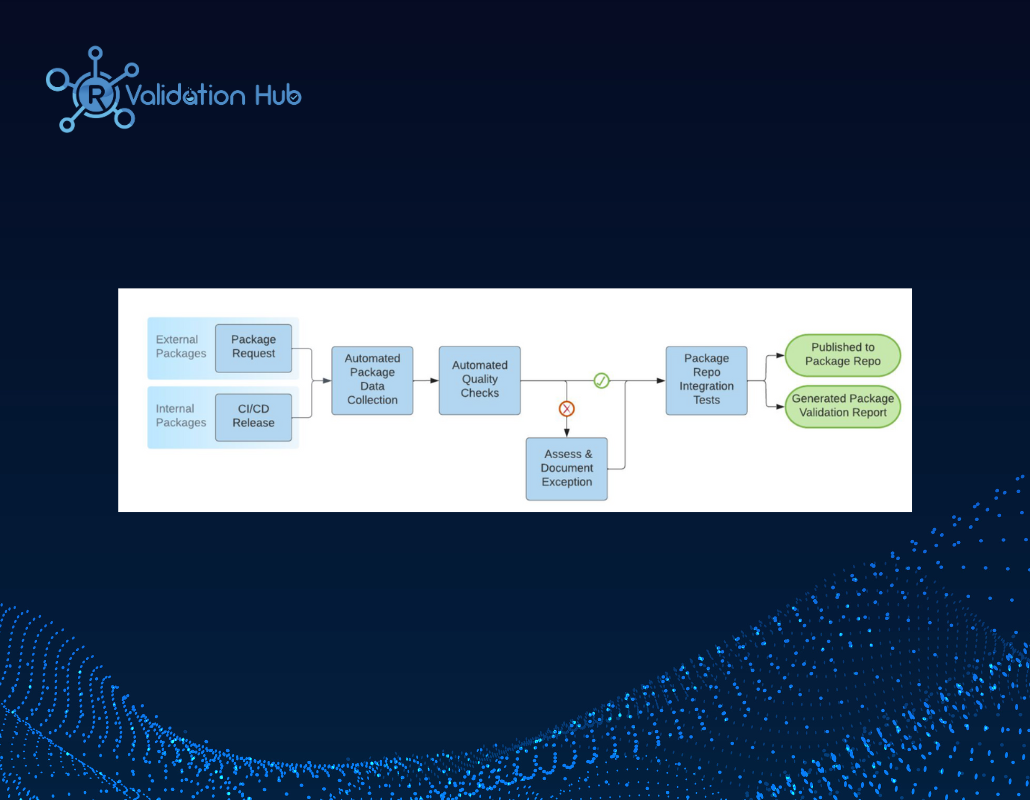
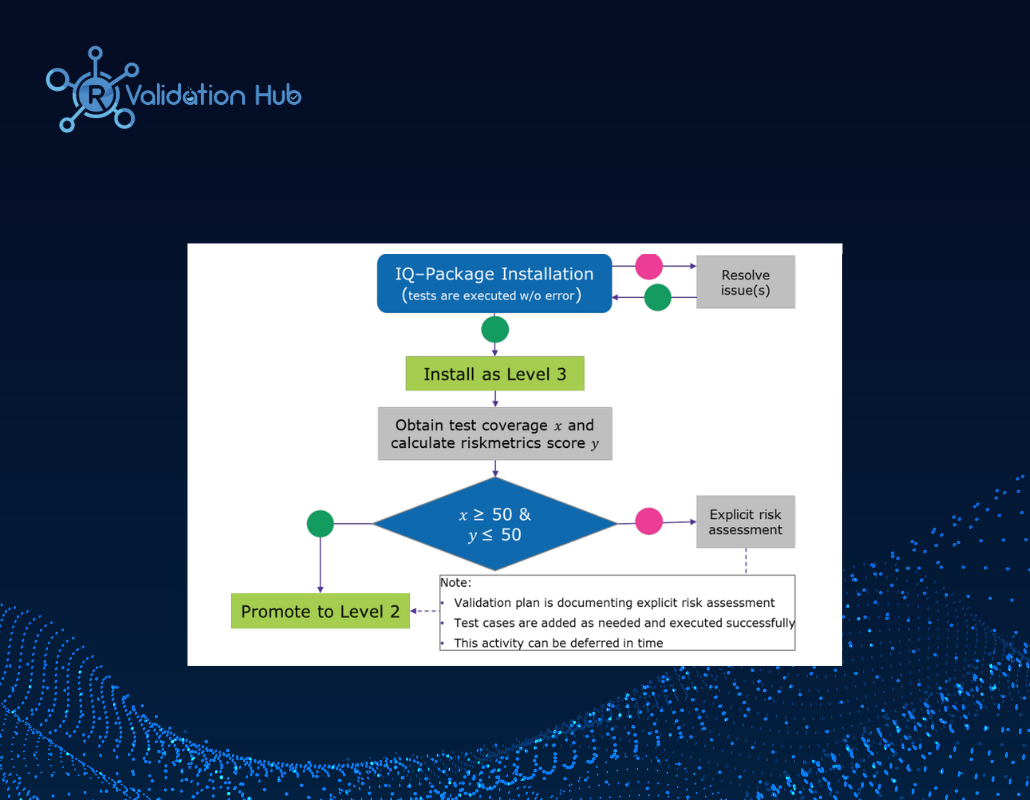
External R Package Qualification Implementation at Merck
Introduction
There has been a growing interest in pharmaceutical industry to use R for clinical trial data analysis and reporting (A&R). Using R for regulatory submission purposes requires careful qualification of R packages given that the open-source packages differ in their quality of development. Many cross-industry initiatives including R Validation Hub and TransCelerate have published framework for qualifying R packages to be used in a regulatory setting (Nicholls, Bargo, & Sims, 2020) (Amoruccio, Lee, & Woodie, 2021). Our organization has been exploring the use of R in a regulatory setting for the past few years. A framework has been developed internally for qualifying external R packages that incorporates elements from both R Validation Hub and TransCelerate framework. This framework is currently being used to qualify both internally developed and externally sourced R packages for use in clinical trial A&R. In this document, we demonstrate this risk-based package qualification framework using the GGally R package. We provide the workflow as well as relevant details regarding the package qualification process used to qualify GGally as a moderate risk R package. We hope this inspires other organizations to use R in a regulatory setting as well as generate discussion to improve our existing framework.